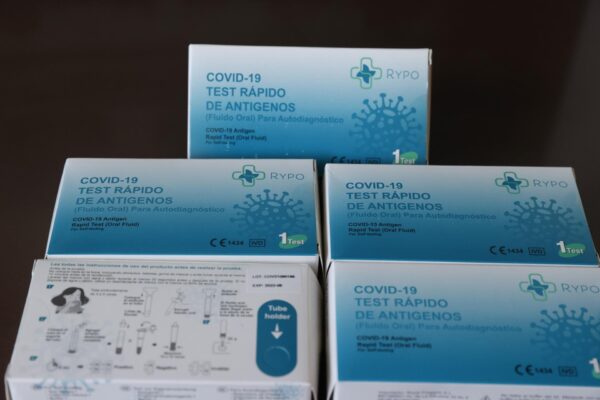
Madrid (EFE).- The Spanish Agency for Medicines and Health Products (AEMPS) has informed this Friday of the cessation of commercialization and withdrawal from the market of all batches of the rapid antigen test for self-diagnosis by saliva ‘Rapid COVID-19’ as it is contaminated by a bacterium the solution in which the swab is dipped.
In addition to requesting the voluntary and definitive cessation of the marketing of the product and the withdrawal of all batches from the Spanish market manufactured by Anbio (Xiamen) Biotechnology co.ltd (China) with an authorized representative in the Netherlands, the Medicines Agency has asked the citizenship that if you have units of these products, do not use them.
According to the AEMPS, the cessation and withdrawal measure of the product is due “to contamination by Pseudomonas aeruginosa of the extraction solution in a new batch 2022012101, with four batches already affected”.
As part of the investigation, the AEMPS has carried out tests in its own laboratory to verify the microbiological quality of the solution from batch 2022012101, detecting levels of microorganisms above those specified in the count tests, as well as the presence of Pseudomonas bacteria. aeruginosa.
The Spanish Medicines Agency has also ordered pharmacies to check if they have units and if so, do not distribute them and withdraw them from sale.
The AEMPS informs that it has learned of the distribution of the product (reference A606201) in Madrid, Seville and Barcelona, although there could be more unidentified distributors.